Tag: bioequivalence
Bioequivalence and Patient Safety: Why Testing Matters for Generic Drugs
- Robin Tudge
- March 5, 2026
- 8 Comments
Bioequivalence testing ensures generic drugs work just like brand-name versions, protecting patient safety while cutting costs. Learn how science, regulation, and real-world data make generics reliable.
read moreBioequivalence Explained: FDA Requirements to Prove Generic Drug Equivalence
- Robin Tudge
- January 8, 2026
- 8 Comments
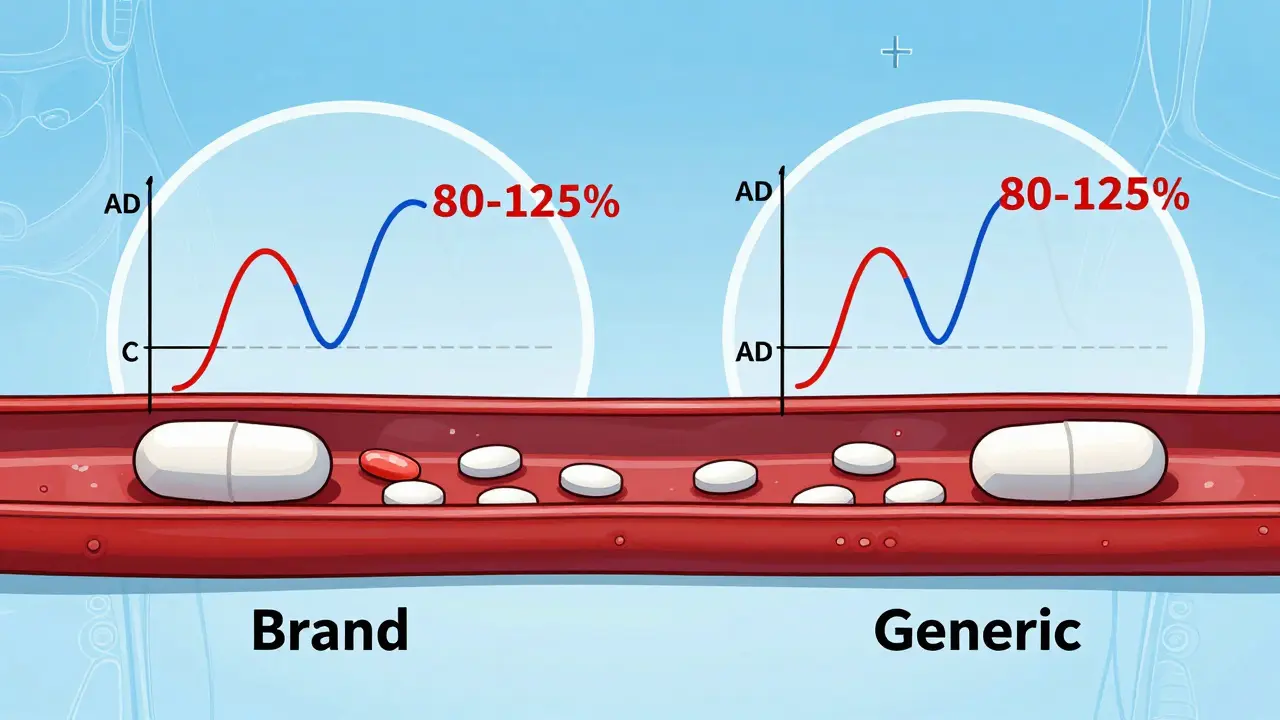
Learn how the FDA ensures generic drugs work just like brand-name ones through bioequivalence testing. Understand the 80-125% rule, what it really means, and why generics are safe and effective.
read moreBioequivalent Medications: What the Term Really Means
- Robin Tudge
- December 12, 2025
- 14 Comments
Bioequivalent medications are generic drugs proven to work the same as brand-name versions in the body. Learn how the FDA tests them, why they're safe, and when to be cautious.
read more